How Does A Fab Fragment Differ From Full IgG Antibodies?
Scientists study antibody structure closely because small differences can lead to major functional changes. Fab fragments and full IgG antibodies share the same origin, yet they behave very differently in biological systems. A full IgG antibody contains both antigen-binding regions and an Fc domain, while a Fab fragment includes only the binding portion. This structural distinction shapes how each interacts with targets, circulates in the body, and triggers immune responses. Researchers do not treat them as interchangeable tools. Instead, they select one based on specific therapeutic or experimental needs. Understanding these differences allows scientists to design more precise strategies in diagnostics, drug development, and targeted therapies.
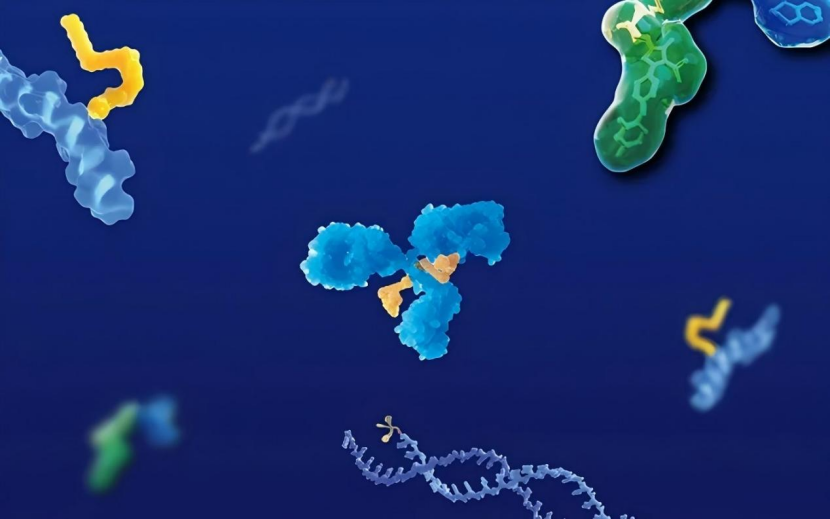
Structural And Functional Differences Between Fab And IgG
Molecular Architecture Defines Their Core Distinction
Scientists begin by examining structure. A full IgG antibody consists of two heavy chains and two light chains, forming a Y-shaped molecule with two antigen-binding sites and one Fc region. The Fc region mediates immune system interactions such as complement activation and receptor binding. In contrast, a Fab fragment represents only one arm of this structure. It contains a single antigen-binding site and lacks the Fc portion entirely. Researchers generate Fab fragments through enzymatic cleavage or recombinant expression. This structural reduction changes how the molecule behaves, making it smaller, more flexible, and less complex in function.
Binding Capabilities Remain But Functional Scope Narrows
Fab fragments retain the ability to bind specific antigens with high affinity. Scientists rely on this property when they need precise targeting without additional immune activation. However, the absence of the Fc region means Fab fragments cannot recruit immune effector functions. Full IgG antibodies, by contrast, can trigger mechanisms such as antibody-dependent cellular cytotoxicity or complement-mediated lysis. Researchers evaluate this difference carefully. When immune activation is beneficial, IgG becomes the preferred format. When selective binding is sufficient, Fab fragments offer a more controlled alternative. This distinction directly influences therapeutic design and experimental outcomes.
Size And Flexibility Influence Molecular Behavior
Scientists also consider physical dimensions. Fab fragments are significantly smaller than full IgG antibodies. This reduced size allows them to penetrate tissues more efficiently, especially in dense or restricted environments such as tumors or inflamed tissues. Full IgG molecules, while larger, benefit from structural stability and longer persistence in circulation. Researchers often observe that Fab fragments distribute more rapidly but clear faster, while IgG antibodies remain in the bloodstream for extended periods. This trade-off between size and stability becomes a central factor when selecting the appropriate antibody format for a given application.
Practical Implications In Research And Therapeutic Applications
Pharmacokinetics Shape Clinical Performance
Scientists analyze how each format behaves over time in the body. Full IgG antibodies interact with neonatal Fc receptors, which protect them from degradation and extend their half-life. This mechanism allows IgG molecules to remain active for days or even weeks. Fab fragments lack this interaction because they do not include the Fc region. As a result, they clear more rapidly through renal filtration. Researchers use this property strategically. In situations where short-term activity is preferred, Fab fragments provide better control. When sustained exposure is required, IgG antibodies offer clear advantages. This difference plays a key role in dosing strategies and treatment design.
Immune System Engagement Varies Significantly
Scientists choose between Fab and IgG based on whether immune activation is desirable. Full IgG antibodies can engage immune cells, amplify responses, and contribute to pathogen clearance or tumor destruction. This makes them suitable for therapies that rely on immune system involvement. Fab fragments, on the other hand, act without triggering these pathways. Researchers use them when they want to block interactions or neutralize targets without causing additional immune effects. Understanding this distinction helps scientists avoid unintended consequences and design safer, more targeted interventions. Development platforms, including those supported by WuXi AppTec, often evaluate these factors early in antibody engineering workflows.
Application Scenarios Drive Format Selection
Scientists match antibody formats to specific use cases. Fab fragments are commonly used in diagnostic imaging, where rapid distribution and clearance improve signal clarity. They are also valuable in situations where immune activation could cause adverse effects. Full IgG antibodies dominate in therapeutic areas that require sustained action and immune engagement. Researchers may also combine formats or engineer hybrid molecules to achieve desired properties. The choice is rarely arbitrary. It reflects a detailed understanding of how structure influences function. The term antibody fragment fab often appears in these discussions as a precise descriptor of a targeted, minimal binding unit designed for specific tasks.
Conclusion
Fab fragments and full IgG antibodies differ in structure, function, and application. Fab fragments consist only of the antigen-binding region, while IgG antibodies include both binding and immune-activating components. This difference shapes how each behaves in the body, from tissue penetration to circulation time and immune engagement. Scientists select between them based on the needs of each project. Fab fragments provide precision and reduced complexity, while IgG antibodies offer durability and functional versatility. By understanding these distinctions, researchers design more effective diagnostic tools and therapies that align with biological and clinical objectives.
Copyright © 2023 guideforests.com. All rights reserved.







